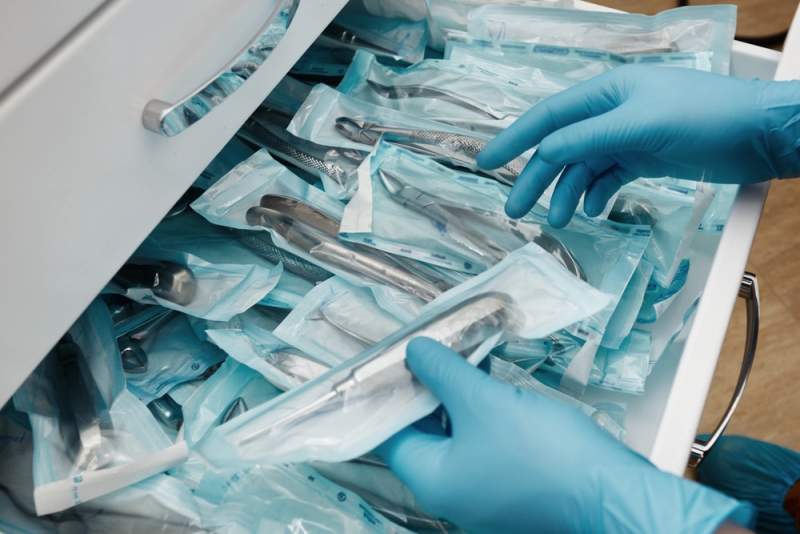

Toray Plastics (America) has introduced its new suite of polyethylene terephthalate (PET) films that can be used in gamma irradiation sterilisation packaging applications.
Featuring three variants, Lumirror GA10, GA25, and GA66, the polyester film range offers improved retention of tensile properties, including tensile strength at break, Young’s modulus, and elongation break.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The films also provide optical properties, such as gloss, haze, and colour, for up to two years after initial radiation exposure by a Cobalt-60 irradiation processor.
Each of the GA films features different functionalities, with GA10 being a printable film, GA25 a heat sealable, while GA66 can be used as a mono web or in packaging laminations.
With better thermal and dimensional stability, the films can be used to supply various medical needs, such as instruments, syringes, implants, catheters, IV sets, surgical gloves, and gauze.
Toray Plastics (America) new business development representative Paul Knollmeyer said: “Toray Industries has been a major supplier of products to the medical, pharmaceutical, and healthcare markets for more than 25 years.
“Our GA Series deepens that involvement and now offers medical-packaging customers an effective, new packaging solution, while presenting converting customers with an opportunity to grow business.”
Listed in the US Food and Drug Administration’s (FDA) drug master file, Toray’s new irradiated GA films can also retain its mechanical properties and can resist damage during storage or handling.
Retention of optical properties ensures clear visibility of the packed product.
It also assures the end user that prolonged storage time has not affected the packaged product and that the item is still sterile and has not been tampered with.
Image: Toray Plastics (America) launches new PET films for medical packaging sector. Photo: courtesy of Toray Plastics (America).





