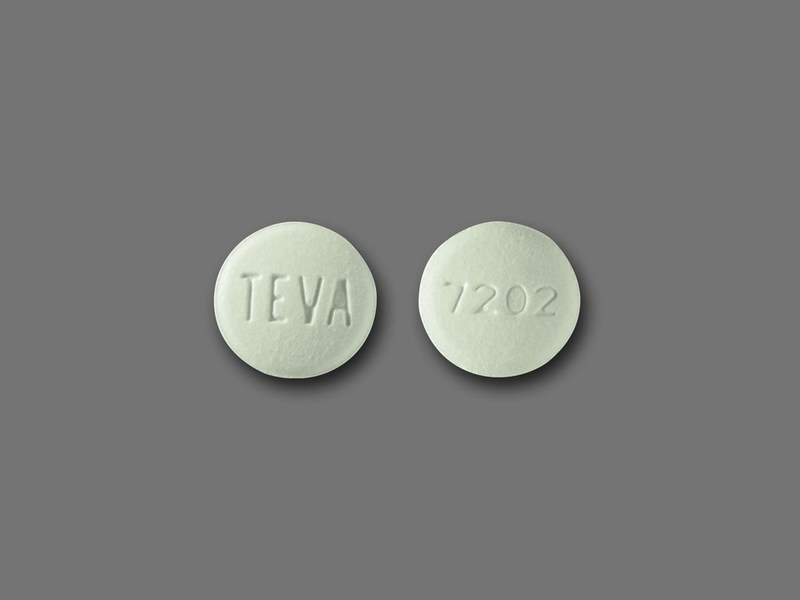

International Laboratories has issued a voluntary nationwide recall of one variant of its Pravastatin Sodium tablets due to mislabelling.
Bottles that have been packaged with the Pravastatin Sodium USP 40mg label reportedly contain Bupropion Hydrochloride XL 300mg tablets.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The company announced the recall after a pharmacist informed that one 30ct bottle of Pravastatin product was mislabelled and contained Bupropion Hydrochloride XL 300mg tablets.
If a patient consumes Bupropion, an aminoketone antidepressant indicated for the treatment of major depressive disorder (MDD), the individual may exhibit various minor side effects, including nausea, vomiting, headache, constipation, sweating and blurry vision.
However, epilepsy patients and people on MAOIs are at a higher risk of possible allergic reactions that can be life-threatening.
The product was delivered to several distribution centres and retail stores in more than 20 US states.
To date, International Laboratories received no complaints or reports of medical illnesses or harmful effects due to mislabelling.
International Laboratories is currently asking its distributors and customers to return all recalled products.
Consumers who have purchased the product will receive a complete refund by returning the sealed package from respective stores or calling the company's complaint number.
Pravastatin Sodium Tablets USP 40mg are an HMG-CoA reductase inhibitor (statin) for patients affected by heterozygous familial hypercholesterolemia.
The company noted that the US Food and Drug Administration was informed about the labelling issue prior to the recall.
Image: Pravastatin Sodium Tablet USP 40 mg. Photo: courtesy of PR NewsFoto / International Laboratories.





